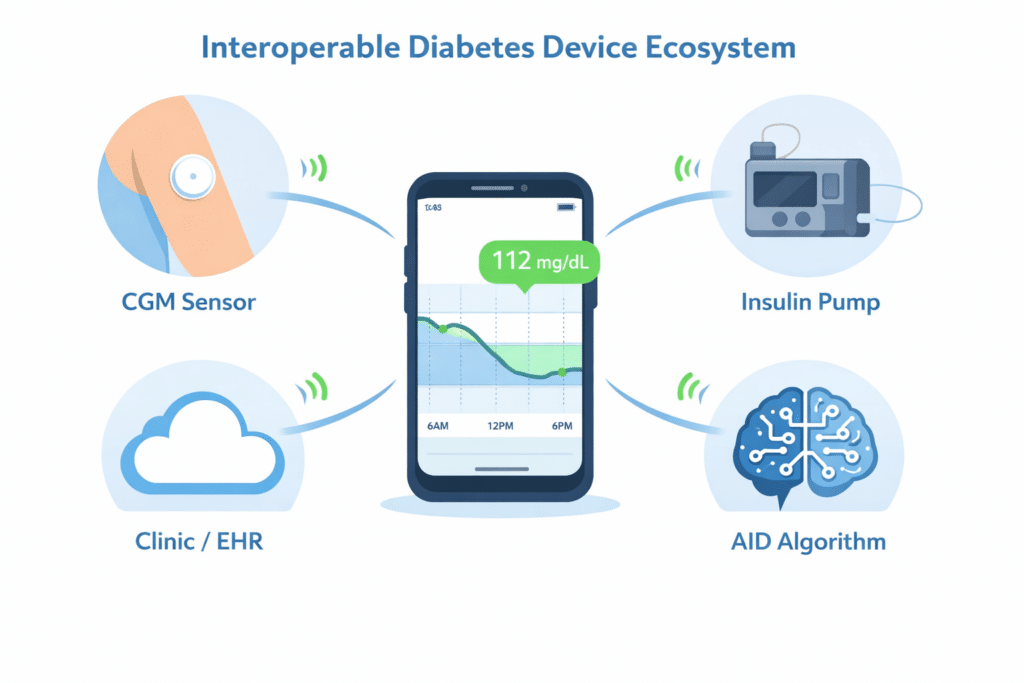
 Interoperable diabetes devices are transforming how you use CGMs, insulin pumps, and automated insulin delivery (AID) systems—allowing parts from different companies to work together safely and reliably. By 2026, interoperability isn’t just a “nice-to-have.” It provides safety for everyday events like sports, illness, puberty, menstruation, and missed meals. It also accelerates diabetes technology development and the speed and quality of your data for your health care team.
Interoperable diabetes devices are transforming how you use CGMs, insulin pumps, and automated insulin delivery (AID) systems—allowing parts from different companies to work together safely and reliably. By 2026, interoperability isn’t just a “nice-to-have.” It provides safety for everyday events like sports, illness, puberty, menstruation, and missed meals. It also accelerates diabetes technology development and the speed and quality of your data for your health care team.
Whether you’re newly diagnosed or a long-time user of diabetes technology, this guide explains what interoperability really means in simple language, how it impacts your daily management, and what to consider when choosing or upgrading your devices.
🔍 Looking for a specific answer? Visit our Frequently Asked Questions for quick info about interoperability in diabetes technology.
Quick Guide: What interoperability really means in everyday life
| Layer | What connects | Why you care |
|---|---|---|
| Local device “talk” | CGM ↔ pump ↔ phone via Bluetooth | Helps dosing stay safe even when you’re busy, active, or asleep |
| Clinical data exchange | Device data ↔ apps ↔ clinic/EHR | Better pattern review, fewer blind spots, smarter dose changes |
| Regulatory interoperability | iCGM + ACE pump + iAGC “plug-and-play” building blocks | More choice, faster upgrades, less “locked-in” hardware |
✅ What’s changed: More AID algorithms have gained interoperable clearance and expanded indications (including for type 2 diabetes). More sensors and pumps are being designed for cross-brand compatibility—so the “mix-and-match” future is getting closer. The arrival of assistance from artificial intelligence is already underway.
What are interoperable diabetes devices?
Interoperability means your diabetes devices can securely share and utilize information—even if they are made by different manufacturers. Practically, it’s the difference between a system that feels “stuck” and one that can grow with you as new CGMs, pumps, and algorithms become available.
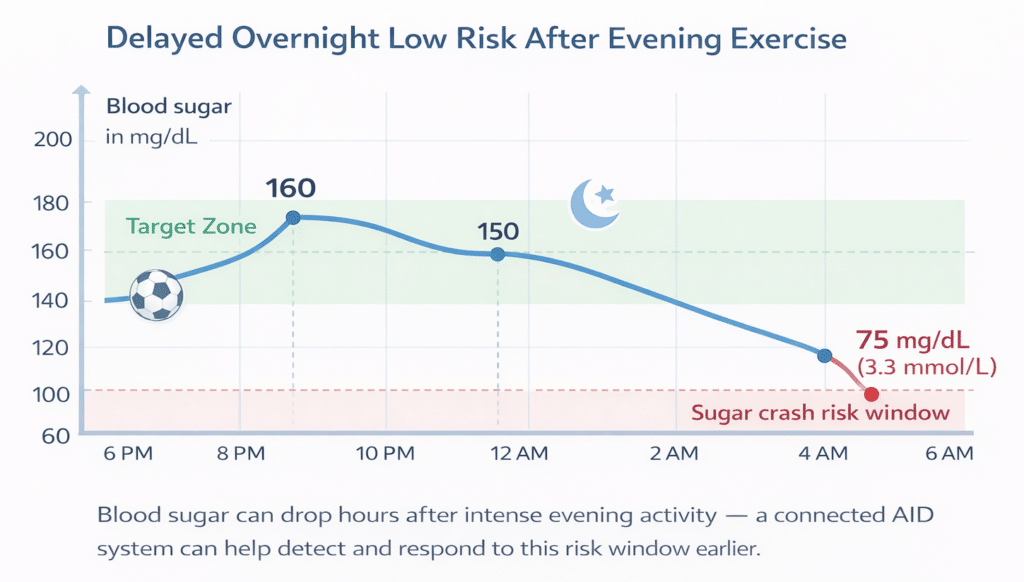
Interoperability is vital when life is unpredictable: growth spurts, puberty, menstruation, illness, stress, travel, and sports can quickly alter insulin needs. When your devices share timely, accurate information—and you have clear backup steps—you can reduce risks like overnight low blood sugar (“sugar crashes”) after exercise or diabetic ketoacidosis (DKA) caused by insulin delivery issues.
How does interoperability enhance safety in real-life situations?
Many dosing problems occur during “rapid change” moments—when your body requires a different plan quickly. Interoperability enhances safety in three essential ways:
- Faster detection: CGM trends enable earlier alerts and smarter AID adjustments.
- Clearer context: Devices can share dosing history, insulin on board (IOB), and device status, so you don’t have to guess.
- Better backup decisions: When something fails, you can pivot quickly because the system shows what happened (or what didn’t).
Example: If your CGM shows 145 mg/dL (8.1 mmol/L) before dinner and you’re heading to a gym or tennis court after dinner, your AID may handle part of the adjustment—but only if connectivity is stable and the system can “see” your CGM trend and insulin delivery history. If a signal drop hides the trend, you might need to step in sooner with a snack, a temporary target, or a manual adjustment.
If you’re new to diabetes technology, start with our simple overview of continuous glucose monitors (CGMs) for a foundation.
How do CGMs, pumps, and AID systems actually communicate with each other?
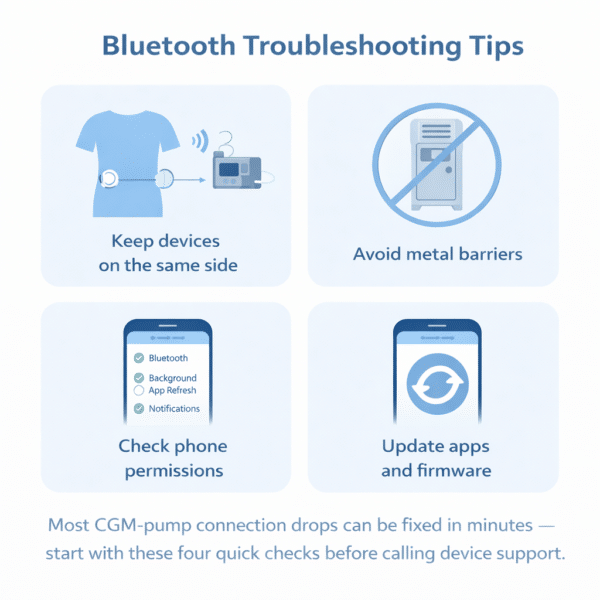
Most modern diabetes devices connect locally via Bluetooth Low Energy (BLE). BLE is energy-efficient and designed for small data bursts, making it ideal for CGM readings and pump commands. However, real-world conditions can interfere: crowded 2.4 GHz environments (such as schools, gyms, and offices), metal barriers (like lockers), and phone settings (background permissions) can cause signal drops. Most of these drops can be fixed with a few simple steps.
 Quick fixes for Bluetooth (BLE) signal drops
Quick fixes for Bluetooth (BLE) signal drops
- Keep devices on the same side of your body: Your body can block BLE signals, especially when you’re moving.
- Avoid metal barriers: Metal lockers, gym equipment, and thick bags can weaken connections.
- Check phone permissions: Ensure Bluetooth, background app refresh, and notification permissions are enabled for your CGM/pump apps.
- Update firmware and apps: Many “mystery disconnects” get better with software updates.
For more background on pump therapy and how modern insulin pumps use CGM data, see our guide to insulin pumps.
What should your health clinic monitor from your devices to keep you safe?
Interoperability isn’t just about your phone. It’s also about whether your care team can access the right details—without overwhelming you with data entry or them with data. A “minimum safety” data set should include:
- CGM quality flags and trends (so the clinic can determine if a data gap is genuine or just a missing signal)
- Insulin dosing history (basal, bolus, corrections) and insulin on board (IOB)
- Time-in-range context and variability indicators
- Basal/bolus balance (helps identify mismatches like “too little basal” versus “missed meal boluses”)
- Meal timing, carbohydrate grams, and, when available, absorption timing
- Activity plus sleep context (especially for exercise-related nighttime lows)
- Device status: battery, reservoir/pod status, site age, occlusion/fault codes
- Situational factors that affect insulin requirements (illness, stress, menstrual cycle timing, rapid growth)
⚠️ Why this matters: Missing or delayed inputs can reduce safety. If your system can’t reliably capture the essentials, you want clear fallback options—manual checks, ketone testing when appropriate, and a written “what to do if…” plan.
If you use an AID system, you’ll also want an easy way to review patterns over time. See our resource on automated insulin delivery systems.
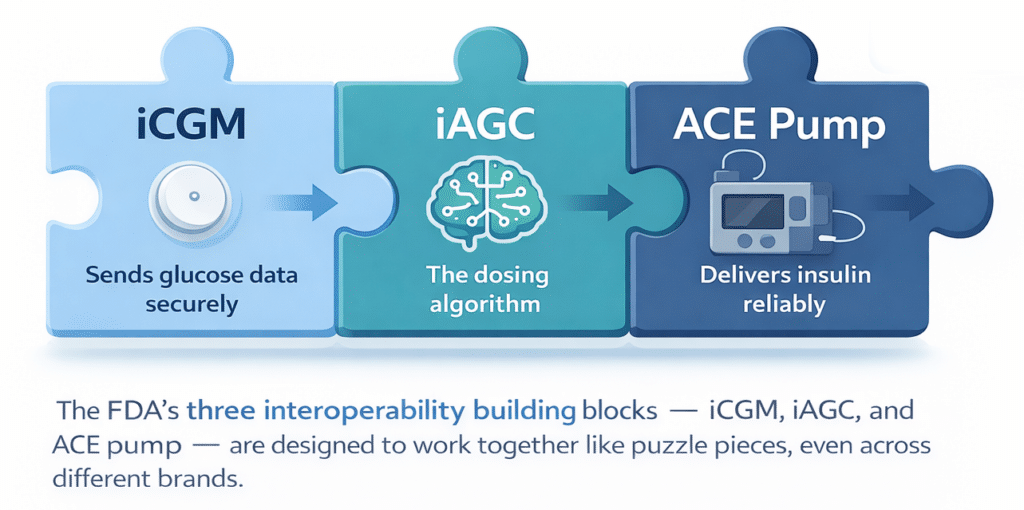
What do iCGM, ACE pump, and iAGC stand for?
The U.S. FDA developed an interoperability framework with regulated building blocks that enable components from different manufacturers to work together more safely.
- iCGM (“integrated CGM”): Engineered to securely transmit your glucose data to connected devices, including AID systems.
- ACE pump (“alternate controller enabled”): Designed to reliably receive dosing commands from a controller or algorithm and confirm that delivery has been completed.
- iAGC (“interoperable automated glycemic controller”): The dosing algorithm, or “brain,” designed to work with compatible iCGMs and ACE pumps in a regulated, plug-and-play manner.
💡 Why it matters: This structure encourages safer, more flexible mix-and-match innovation. Instead of replacing everything at once, you can potentially upgrade one part (like a CGM) while keeping another part (like a pump)—as long as the combination is validated and listed as compatible.
How has interoperability in diabetes technology evolved?
2010: The introduction of Bluetooth Low Energy (BLE) introduced a crucial technology for reliable, low-power device communication. Think of BLE as the “short, frequent check-in” signal that helps your CGM and pump stay synchronized without draining batteries.
2017: JDRF (now known as Breakthrough T1D) launched the Open Protocols Initiative to promote “plug-and-play” compatibility for AID components—aiming to increase choices and reduce lock-in.
2018–2019: The FDA approved key interoperability milestones, including the first iCGM designation (Dexcom G6), the first ACE pump designation (Tandem t:slim X2), and initial interoperable controller pathways, helping normalize connected AID systems.
2025–2026: Several controllers and algorithms have expanded capabilities and broader indications, including for type 2 diabetes. More CGMs are being evaluated for iCGM performance expectations, and more companies are building ecosystems that anticipate the integration of multiple sensors and algorithms over time.
What does the growth of interoperability mean for you?
Recent significant changes in diabetes device interoperability:
- More iAGC momentum and broader indications: Several controllers and algorithms have expanded capabilities, signaling that interoperable AID is moving beyond a single “one-size-fits-all” audience.
- More sensor options designed for integration: Newer CGMs have been evaluated in the context of iCGM performance and integration expectations.
- More partnerships: Companies are increasingly developing ecosystems that expect the integration of multiple sensors and algorithms over time. Activity monitors and hormone/chemistry monitoring are becoming more common.
Bottom line: When you evaluate a new pump, CGM, or AID system, don’t just ask “Is it good?” Instead, ask: “How easily can I upgrade one part later? How quickly will new technology be integrated? What happens during failures? What new sensing will be available?”
Why is reliable insulin delivery a key topic for interoperability?
Interoperability isn’t just about data sharing; it also involves detecting “silent failures” in insulin delivery. Failures in infusion sets and adhesives can raise blood sugar levels and, in some cases, lead to DKA. A connected ecosystem assists by standardizing device status signals in apps and portals: battery level, reservoir or pod status, site age, fault codes, and occlusion alerts.
One practical observation: many pump users notice their average blood sugar tends to rise as infusion set wear time increases. A smart system could track “average glucose by site-day” and help you identify when you’re more likely to run high on day 3 or 4 compared to day 1—so you can adjust or change sites sooner.
⚠️ Safety reminder: If you have persistent high readings, such as 250 mg/dL (13.9 mmol/L), with symptoms like nausea, stomach pain, deep breathing, or unusual fatigue—or if you suspect insulin delivery failure—follow your clinician’s sick-day plan and perform ketone testing. Don’t wait. An AID can help identify, but cannot fix a delivery problem.
Interoperability enables early alerts when multiple signals are combined—especially in two high-risk scenarios:
2) Early identification of DKA risk
DKA often begins with insulin interruption and increasing ketone levels. Interoperability can reduce risk when systems can “see” both:
- Glucose trends and insulin delivery status (site or pod issues, occlusions, missed boluses)
- Ketone level (when available) to help identify the rise in ketone levels earlier
3) Menstrual cycle and hormonal fluctuations
For women with diabetes, the menstrual cycle significantly impacts blood sugar levels each month. An increase in estrogen often signals the start of menstruation, while a decrease in estrogen and an increase in progesterone mark its end. During menopause, the ovaries produce much less estrogen and progesterone, leading to symptoms like hot flashes, mood swings, and sleep issues—all of which can influence blood sugar control. Monitoring these hormone levels with sensors could greatly improve insulin delivery estimates.
Education remains a critical factor—many people with type 1 diabetes have never experienced DKA and may disregard early symptoms. Connected systems can assist, but they can’t replace understanding and planning for DKA.
4) Stress and cortisol
Most people using a CGM know that stress greatly influences blood sugar. Cortisol is the primary hormone regulating daily stress levels. A combined glucose and cortisol sensor could significantly improve blood sugar management on a daily basis for some, and boost awareness of one’s overall health and the benfits of stress management.
The future: sensing beyond glucose
Glucose is just one part of your physiology. Upcoming step s include multi-signal monitoring and integrating glucose with additional signals to enhance accuracy and minimize false alarms. Examples include:
- Ketones for early DKA risk detection
- Activity intensity (heart rate zones/METs) to enhance prediction of exercise-related lows
- Temperature and heart-rate variability (HRV) to detect illness or stress changes
- Hydration context to understand unusual trends
The clinical promise is real and relies on interoperability: sensors and devices that share additional signals reliably that reach you when you need them most.
How does AI fit in—and why does “on-device” intelligence matter?
As diabetes technology advances, it must also become more transparent. A few practical principles help ensure AI remains useful:
- Prioritize local for safety: Your essential therapy should remain accessible even if the internet goes down.
- Edge AI when offline: On-device inference can enable alerts and pattern detection during outages.
- Cloud for learning and care coordination: Clinics and telemedicine benefit when summarized insights are transmitted back to portals and electronic health records (EHRs).
Request transparency: As AI features grow, it’s reasonable to ask vendors for straightforward “model cards” that detail what the feature does, what it doesn’t do, how recently it was validated, and what could make it less reliable.
 What questions should you ask before selecting an interoperable setup?
What questions should you ask before selecting an interoperable setup?
Whether you’re newly diagnosed or have used tech for years, these questions can help prevent frustration and lower risks:
- Compatibility: “Which CGMs/pumps/controllers are currently officially supported together?”
- Disconnect behavior: “If Bluetooth drops, what changes? What stays safe? What alerts will I see?”
- Data quality: “Do you label missing or low-quality CGM data clearly so I don’t overreact?”
- Device status visibility: “Can I easily see battery, reservoir/pod status, site age, and fault codes?”
- Upgrade path: “If I want to upgrade my CGM next year, will I have to replace my pump too?”
If you want a deeper understanding of how dosing decisions are made in real life—including meals, insulin timing, and activity—our resources on continuous glucose monitoring, insulin pumps, and automated insulin delivery can help you build confidence step-by-step.
Interoperability standards
Interoperability relies on a mix of standards and policy frameworks:
| Category | Examples | Plain-language meaning |
|---|---|---|
| Clinical data exchange | HL7 FHIR, SMART, USCDI, TEFCA | How your health data moves between apps, portals, and EHRs |
| Device communication | Bluetooth profiles, IEEE device standards | How devices share readings/commands locally and reliably |
| FDA interoperability building blocks | iCGM, ACE pump, iAGC | Regulatory “plug-and-play” categories for AID components |
A helpful tip: When selecting new technology, ask the manufacturer for (1) compatibility lists and (2) information about disconnects: “Does the system keep safe insulin delivery locally? What alerts will I see? What should I do next?”
Helpful Resources & Research
- ADA Standards of Care 2026: Section 7 (Diabetes Technology)
- FDA: First device to enable automated insulin dosing for type 2
- eCFR: FDA definition for integrated continuous glucose monitoring (iCGM)
- FDA classification: Alternate Controller Enabled (ACE) insulin pump
- FDA 510(k) summary: Interoperable controller (Control-IQ+)
Frequently Asked Questions
What are interoperable diabetes devices?
Interoperable diabetes devices include CGMs, pumps, and AID systems that can safely share data—even across different brands. This makes it easier to upgrade one part of your setup over time while keeping the rest the same.
What does iCGM stand for in simple language?
iCGM means an “integrated CGM” that’s designed to securely send your blood sugar readings to other devices like insulin pumps and AID systems. It’s intended to support safer connections, not just display numbers on your phone.
What is an ACE pump, and why is it important?
An ACE pump is an insulin pump designed to dependably receive dosing commands from a controller or algorithm and to verify that delivery has taken place. Safe automation depends on both the command and the confirmation—especially during overnight use.
What is an iAGC in an AID system?
iAGC stands for interoperable automated glycemic controller—essentially the “brain” (algorithm) that uses your blood sugar trends to automatically adjust insulin. It’s designed to work with compatible iCGMs and ACE pumps in a regulated, plug-and-play manner.
Is it possible to mix and match CGMs and pumps from different companies?
Sometimes—if that specific combination is listed as compatible by the manufacturers and supported by regulatory clearance. Always check official compatibility lists, as they change with new updates and devices.
What should I do if my CGM and pump keep losing Bluetooth connection?
First, try simple fixes: keep devices on the same side of your body, avoid metal barriers like lockers, and make sure your phone’s Bluetooth and background app permissions are enabled. If disconnects persist, update firmware and apps, then contact the device support line—frequent dropouts can compromise your safety.
How can interoperability lower my risk of DKA?
Interoperability can enhance visibility of insulin delivery status—like occlusions, site or pod issues, and missed doses—making it easier to respond quickly. If you notice sustained high blood sugar such as 250 mg/dL (13.9 mmol/L) and suspect a delivery failure, follow your sick-day plan and consider testing for ketones.
How can interoperability help prevent overnight sugar crashes after exercise?
Connected systems can analyze your blood sugar trends, insulin history, and activity signals to identify higher-risk periods after intense workouts. Some people find it helpful to set a higher overnight target or plan a snack when exercise increases the risk of a late low.
What data should my clinic ideally see from my diabetes devices?
The most valuable data usually includes blood sugar trends with quality flags, insulin dosing history, insulin on board (IOB), meal timing and carbohydrate entries, and device status (battery, reservoir or pod, site age, fault codes). This helps your care team focus on “what’s next” instead of guessing what happened.
Is “more data” always better in diabetes technology?
Not always. Better data is timely, accurate, and clearly labeled—especially when something is missing or unreliable. The goal is to have fewer blind spots and clearer next steps, not to generate more graphs that leave you guessing.
Last Updated on February 26, 2026